- One of the most remarkable medical findings in the new
21st century is the recognition that 90% of the cells that make up our
body are not human cells. On the contrary, most of our cells are the trillions
of microbial cells (primarily bacteria) that inhabit our bodies and share
our lives. According to microbiologist Brett Finlay: "We really are
a superorganism. From the moment we are born until we die, we live in a
symbiotic relationship with our microbes."
-
- The National Institutes of Health has launched a $115-million
Human Microbiome Project to identify, analyze and catalog the hundreds
of microbial species residing in or on the human body. Modelled after the
Human Genome Project, which decoded most of the human genes in the 1990s,
the goal of the microbiome project is to determine which microbes are harmful
and to figure out ways to prevent or treat diseases they cause.
-
- "Pleomorphic" bacteria and human cancer
-
- It is peculiar that the trillions of bacteria that are
part of us do not appear in textbooks of histology (the microscopic study
of normal tissue) and pathology. And there is no pathology textbook of
cancer that illustrates bacteria in cancerous tissue. This is strange because
bacteria, even with their small size, can be seen with a light microscope,
unlike viruses which cannot be visualized microscopically due to their
smaller size. If human bacteria were implicated in the production of human
cancer, they would undoubtedly be visible microscopically in cancer tissue.
This report suggests that bacteria are indeed implicated in cancer, and
that they can be seen in cancerous tissue.
-
- The idea that bacteria might be connected in some way
to cancer was totally rejected a century ago by the medical establishment.
Since that time any association between bacteria and cancer has been largely
regarded as medical heresy. Nevertheless, a few foolhardy investigators
(including myself) have continued to report on pleomorphic bacteria as
a possible cause of cancer.
-
- "Pleomorphism" means the ability of a microbe
to exhibit more than one forms. For more than a half-century, bacteria
associated with cancer have been described as having a "life cycle"
comprised of virus-sized forms, filterable granules, larger granules the
size of ordinary cocci, "globoidal forms," rod-like forms, much
larger fungus-like forms, and other forms. To traditional microbiogists
who believe in the "monomorphic" nature of bacteria, that is
the ability of a microbe to reproduce by merely splitting in half, the
idea of a cancer germ with a life cycle is considered anathema. Despite
this tremendous controversy, the proposed microbiology of cancer cannot
be understood without considering so-called pleomorphic bacteria. Highly
recommended is microbiologist Milton Wainwright's "Extreme pleomorphism
and the bacterial life cycle: A forgotten controversy," available
on the Internet.
-
- Despite the widespread rejection of the bacterial theory
of cancer, Australian researchers Barry Marshall and Robert Warren discovered
in 1982 that bacteria (now called Helicobacter pylori ) were frequently
the cause of stomach inflammation (gastritis) and stomach ulcers. They
were awarded the Nobel Prize in Medicine in 2005 for this pioneering work.
Ulcers can sometimes lead to stomach cancer; and doctors now accept H.
pylori as a bona fide cause of this type of cancer. Prior to the acceptance
of bacteria-caused ulcers and cancer, it was firmly believed that bacteria
could not exist and thrive in the acid environment of the stomach. H. pylori
can now be easily identified microscopically, provided a special stain
(like a silver or a Giemsa stain) is applied to the stomach biopsy tissue.
The microbe is pleomorphic, having both a spiral and a coccoid intracellular
form.
-
- Cancer bacteria research
-
- Despite the general non-acceptance of a bacterial cause
of cancer, there is a wealth of research dating back to the late nineteenth
century on this subject. The most important contributions to the bacteriology
of cancer were reported by a team of four women, beginning in the 1950s.
These included physician Virginia Livingston, microbiologist Eleanor Alexander-Jackson,
cell cytologist Irene Diller and biochemist and tuberculosis icon Florence
Seibert.
-
- Livingston's cancer work actually began in 1947 when
she discovered tuberculosis-like acid-fast bacteria in a non-cancerous
connective tissue disease called scleroderma, a systemic disease in which
the skin hardens due to thickening and abnormalities of the collagen portion
of the skin. This scleroderma research quickly led her and her co-workers
into cancer research in which similar acid-fast bacteria were found and
studied extensively over the next three decades. Livingston believed the
cancer microbe was essentially a "connective tissue parasite."
In scleroderma tissue the microbes are seen in their most naked form infiltrating
the collagen fibers and bundles (Fig.6)
-
- My mentors Livingston and Alexander-Jackson were the
first to discover that bacteria could be detected in cancer biopsy material
if a special "acid-fast" stain was applied to the tissue specimens.
The women also realized that cancer bacteria survived in the body in the
"cell wall deficient form", thus enabling these microbes to escape
detection and/or eradication by the immune system. Such bacteria were derived
from "ubiquitous bacteria" normally residing in all human beings.
-
- Various researchers have reported the cancer germ as
an intracellular and an extracellular parasite, meaning that the bacteria
can be found both within and outside the cell. The shape of the bacteria
is usually the round coccus-like form. These coccal forms vary in size
from the size of ordinary staphylococci to extremely tiny granular forms
barely visible with the light microscope. These forms in tissue are compatible
with pleomorphic forms of cell wall deficient bacteria. Some CWD bacterial
forms have the ability to pass through laboratory filters designed to hold
back bacteria. Thus, some growth forms of cancer bacteria are virus-sized
and submicroscopic. The relationship, if any, of these submicroscopic virus-like
forms of CWD cancer bacteria to the "true" viruses has not been
studied.
-
- Cancer cytologist Irene Diller and her co-workers demonstrated
that these intracellular bacteria could also be found within the nucleus
of the cell. Seibert stressed that this was an extremely important finding
because it indicated these bacteria could have access to the genetic information
stored within the cell nucleus, thereby inducing cancerous cell changes
via transfer of genetic information.
-
- The appearance of bacteria in cancerous tissue
-
- It is not possible to easily visualize cancer bacteria
in the routine tissue stain (hematolylin-eosin stain) employed to diagnose
cancer. (This was also the reason that stomach bacteria were never detected
in ulcers and stomach cancer.) Although most physicians cringe at the idea
of a "cancer microbe", such bacteria can be seen in cancerous
tissue sections by using the highest magnification of the light microscope
and oil immersion, as well as a suitable stain like the "acid-fast"
stain. The acid-fast stain is a time-honored stain used to detect acid-fast
(red-stained) forms of tuberculosis (Tb) bacteria.
-
- In addition to the typical purple-stained or blue-stained
intra and extracellular coccoid forms characteristic of the cancer microbe,
one can occasionally encounter large bizarre forms of pleomorphic cell-wall-deficient
bacteria in tissue sections. Such forms are known to microbiologists as
"large bodies." They can attain the size of red blood cells and
even larger. These may be the forms described in cancer tissue in the late
nineteenth century by Scottish pathologist William Russell, who termed
such elements as "the parasite of cancer." (For more details
on "Russell bodies" and "large bodies" and their implications
in the cancer process, see my internet article "The Russell body:
The forgotten clue to the bacterial cause of cancer".)
-
- A century ago microbiology was in its infancy and cell
wall deficient bacteria and "large bodies" were unknown. But
experts nevertheless totally discredited Russell's "parasites"
as mere cellular degenerations of no great import. His so-called cancer
parasites are now widely known by pathologists as "Russell bodies,"
who consider them as non-microbial in nature. The possibility that they
could represent pleomorphic forms of bacteria is never considered. Various
recent reports confirm the existence of a new pathologic condition of the
stomach, associated with Helicobacter pylori infection and numerous Russell
bodies and Russell body-containing plasma cells. The new entity is called
"Russell body gastritis."
-
- Physicians often argue that any bacteria associated with
cancer are merely "secondary invaders"-bacteria which can easily
proliferate in cancer-damaged tissue after the cancer has formed. However,
a careful study of "pre-cancerous" lesions, such as sun-damaged
skin, will reveal that the bacteria are present prior to the development
of full-blown skin cancer. Additionally, a study of breast cancer tissue
supposedly "clear" of cancer cells can show the presence of these
bacteria in cancer-free tissue areas. One of my reported patients with
Hodgkin's disease (lymphoma) was cured of his cancer, but the bacteria
that were associated with his cancer were still present at autopsy, indicating
that radiation does not kill human bacteria, although it can destroy cancer
cells and eliminate cancer tumors. It is likely that these primordial bacteria
that have grown side by side with human cells for eons will prove to be
the hardiest life forms on earth, perhaps even achieving immortality.
-
- The purpose of this brief report is to remind people
interested in the cause of cancer that bacteria have been detected and
reported in major forms of cancer, such as breast cancer, prostate cancer,
AIDS-related Kaposi's sarcoma, lung cancer, Hodgkin's lymphoma, as well
as in certain other cancerous and non-cancerous diseases. Photos of this
cancerous tissue (Figures 1-5) illustrate the appearance of bacteria encountered
in acid-fast stained tissue sections. Figure 6 shows these coccoid forms
most clearly in the dermis portion of skin in scleroderma, the disease
which originally led to Virginia Livingston's discovery of the "cancer
microbe." Whether bacteria can be demonstrated in every form of cancer
and in every patient from whom cancerous tissue is available, is not known.
-
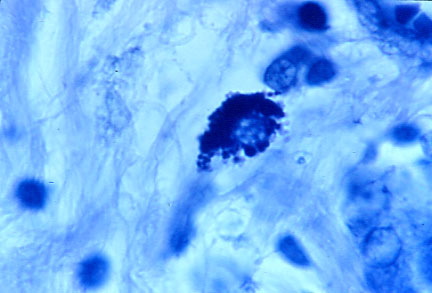
-
- Figure 1: In center, intracellular round tightly-packed
coccoid forms in breast cancer. Acid fast stain, magnification times 1000,
in oil.
-
-
-
- Figure 2. Intracellular coccoid forms in prostate
cancer. Acid-fast stain, x1000, in oil.
-
-
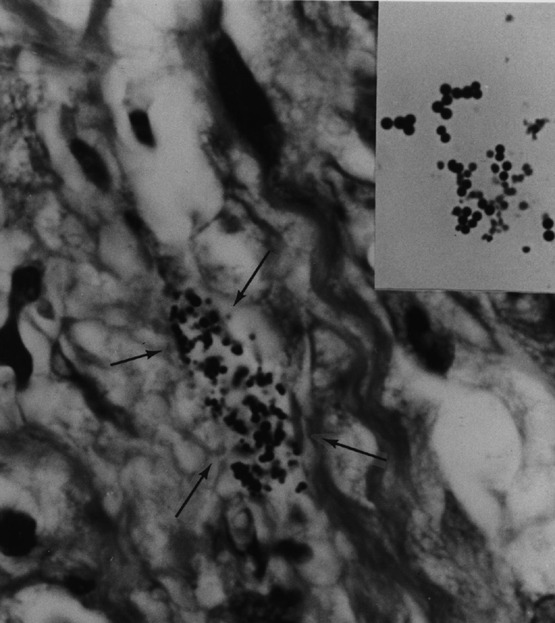
-
- Figure 3. Arrows point to extracellular coccoid
forms in the dermis of the skin of AIDS-related Kaposi's sarcoma. In the
insert are staphylococci cultured from the tumor. Acid-fast stain, x1000,
in oil.
-
-
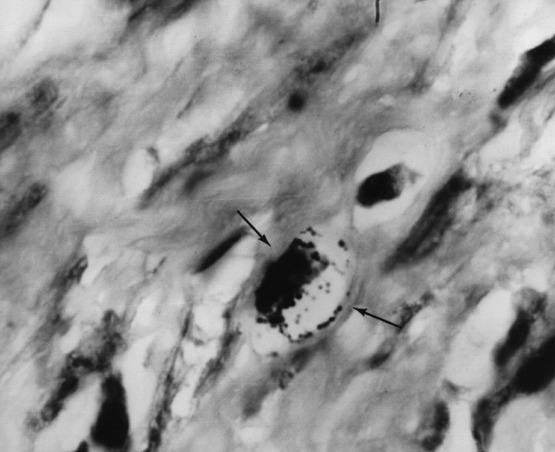
-
- Figure 4. Arrows point to intracellular and extracellular
coccoid forms in the lymph node showing Hodgkin's lymphoma. Acid-fast stain,
x1000, in oil.
-
-
-
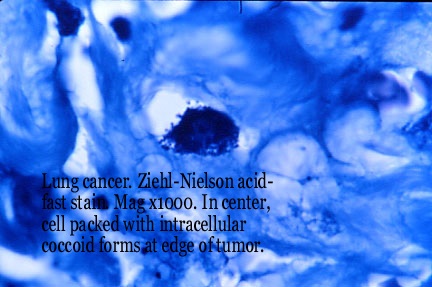
-
- Figure 5. Intracellular tightly-packed coccoid
forms in lung cancer at the edge of the tumor. Acid-fast stain, x1000,
in oil.
-
-
-
-
-
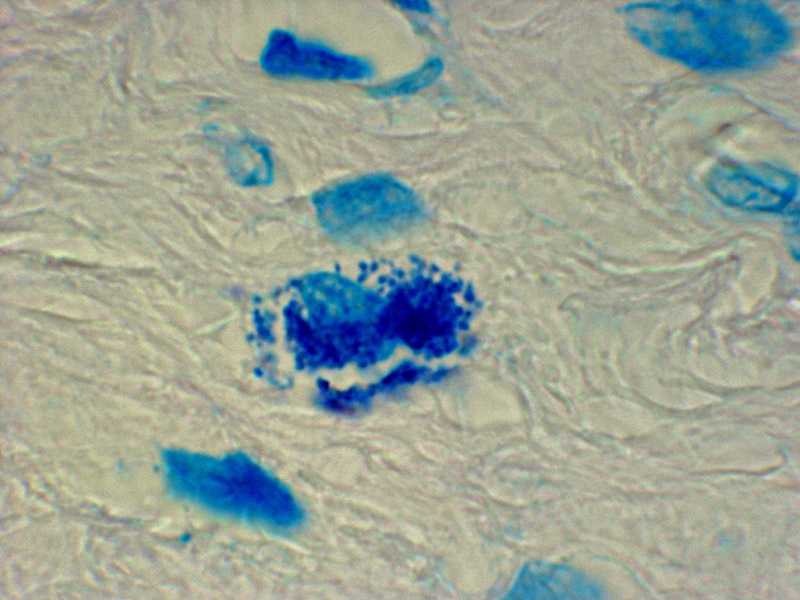
- Figure 6. "Naked" coccoid forms nestled
between the collagen fibers of the skin of scleroderma. Acid-fast stain,
x1000, in oil.
-
-
-
- New developments in 21st century microbiology
-
- The discovery of trillions of "human bacteria"
should lead to renewed interest in their role as possible cancer-producing
agents and agents of chronic disease. The Human Microbiome Project (HMP)
is currently characterizing these communities of bacteria (called the "metagenome")
and studying their collective influence on human development, physiology,
immunity, and nutrition. As a result, new insights into the production
of disease are beginning to emerge. Proal and co-workers (2009) in their
study of bacteria in chronic autoimmune disease write: "As our knowledge
of the genes associated with the disease states expands, we understand
that humans must be viewed as superorganisms in which a plethora of bacterial
genomes - a metagenome - work in tandem with our own." Some of the
90% of microbial cells in Homo sapiens create metabolites that interfere
with the expression of genes associated with autoimmune disease.
-
- More and more research suggests that bacteria share and
exchange genes with one another in a process known as "horizontal
transfer of genes." Can bacteria transfer their genes to human cells?
Could transfer of bacterial genes to human cells induce cancerous change?
-
- In this regard, Agrobacterium tumefasciens is a bacterium
that produces tumors (crown gall) in plants by inserting a bit of its DNA
into the plant cell. The Agrobacterium host range is not limited to the
plant kingdom; the microbe has been shown to also transform many species
of fungi and even prokaryotes (cells that lack a nucleus). The ability
of Agrobacterium to genetically transform human-derived cancerous HeLa
cells further widens the range of potential hosts of Agrobacterium to include
humans and perhaps other animal species (Tzfira et al. 2006).
-
- One of the century-old stumbling blocks causing widespread
rejection of a bacterial cause of cancer has also been the inability of
researchers to precisely identify the type and species of so-called cancer
microbes. The intermittent acid-fastness of cancer bacteria suggests a
close relationship to the mycobacteria, the most important species of which
is Mycobacterium tuberculosis, the cause of human Tb. However, in general,
bacteria cultured from cancer are highly pleomorphic and are impossible
to classify precisely. This is also consistent with CWD bacteria found
universally in the blood of both healthy and diseased individuals. Such
blood bacteria have been shown to be a hodge-podge of staphylococcal and
streptococcal-like bacteria, as well as cocco-bacilli.
-
- The inability to precisely characterize cancer germs
is also in keeping with the current revolution in microbiologic thought
regarding the classification of bacteria. In a recent essay in Nature,
Goldenfeld and Woese (2007) state that the "emerging picture of microbes
as gene-swapping collectives demands a revision of such concepts as organism,
species and evolution itself. The available studies strongly indicate that
microbes absorb and discard genes as needed, and in response to their environment.
Rather than a discrete spectrum, we see a continuum of genomic possibilities,
casting doubt on the validity of the fundamental concept of species, extended
into the microbial realm. The lack of usefulness of the species concept
is inherent in the recent forays into metagenomics."
-
- Bacteria and "the cause of cancer"
-
- If one googles "the cause of cancer", the American
Cancer Society website provides a laundry list of well-accepted causes,
including the most obvious: smoking, diet, radiation, excessive sun exposure,
environmental carcinogens, inherited genetic abnormalities, to name only
a few. Human bacteria of the kind discussed and illustrated in this report
are nowhere on the list. This is because they are not taken seriously by
the medical establishment. Like the bacteria in stomach ulcers that were
unrecognized for a century, these bacteria comprise the best kept secret
in medical science.
-
- How could unrecognized bacteria cause cancer? All cancer-causing
agents injure tissue and cells. Radiation, for example, can also transform
the genetic structure of cells. And human bacteria could easily proliferate
in damaged pre-cancerous tissue. In addition, inflammation has long been
associated with cancer; and bacteria are frequently associated with inflammation.
It seems reasonable to assume that the trillions of bacteria in our bodies
could play some role in the abnormal tissue and cellular environment that
precedes the development of cancer.
-
- It is hoped that this communication will stimulate further
thought, open mindedness, and investigation into the role of bacteria in
human cancer. The century-old experience with undetected stomach bacteria
indicates the dangers of dogmatism in medical thought. It is possible that
a recognition of these primordial and human bacteria will lead to new ways
to prevent and treat cancer by eliminating and/or lessening the proliferation
of these microbes in diseased tissue.
-
- The idea that "human bacteria" might play a
role in cancer is both sobering and frightening. How do we restore healing
when our own germs "act-up"? However, knowledge of these bacteria
is essential if we are to deal with this reality. As Walt Kelly of Pogo
fame once wrote: "We have met the enemy and he is us." Sobering
indeed!
-
- ***
-
- This communication is dedicated to the memory of Lucia
Cabrera, R.N. [1968-2011], cancer victim.
-
- Alan Cantwell is a retired dermatologist who has studied
the microbiology of cancer for over 40 years. He is the author of "The
Cancer Microbe" and "Four Women Against Cancer: Bacteria, Cancer,
and the Origin of Life." His Internet writings can be found by Googling:
alan cantwell articles. His scientific papers are recorded at the PubMed
website (Key word: Cantwell AR). For a bibliography of cancer microbe research,
email: alancantwell@sbcglobal.net.
|





